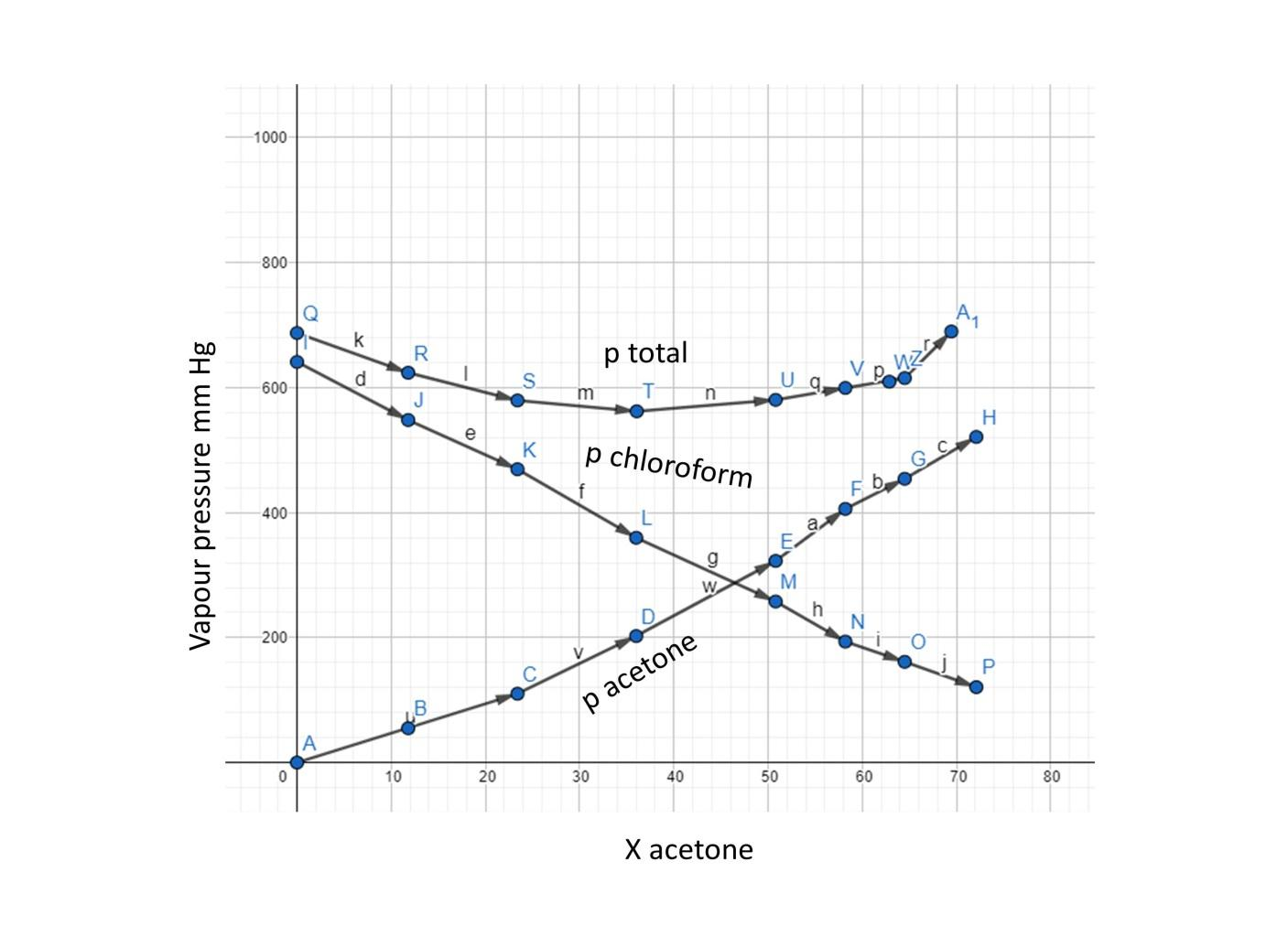
Vapour pressure of pure acetone and chloroform at 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of composition, plot

SOLVED:The vapor pressure of acetone (CH3 COCH3) at 19^∘ C is 5.33 kPa. A 3.50-g sample of acetone is placed in a 2.00 L container at 22^∘ C. If the container is

QUESTION 2.37 Vapour pressures of pure acetone and chloroform at 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of
The vapour pressure of acetone at 20°C is 185 torr. When 1.2 g of a non-volatile substance was dissolved in 100 g of acetone at 20°C, - Sarthaks eConnect | Largest Online Education Community

SOLVED: Vapor pressures and vapor compositions for Acetone-Chloroform solution at 25°C. cclu (20)- shnbm^ acedne 332 Xc PclkPa PakPa 46.7 0.0 0.0 46.3 0.2 4.7 42 33.3 8 Fak 0.4 11.0 4